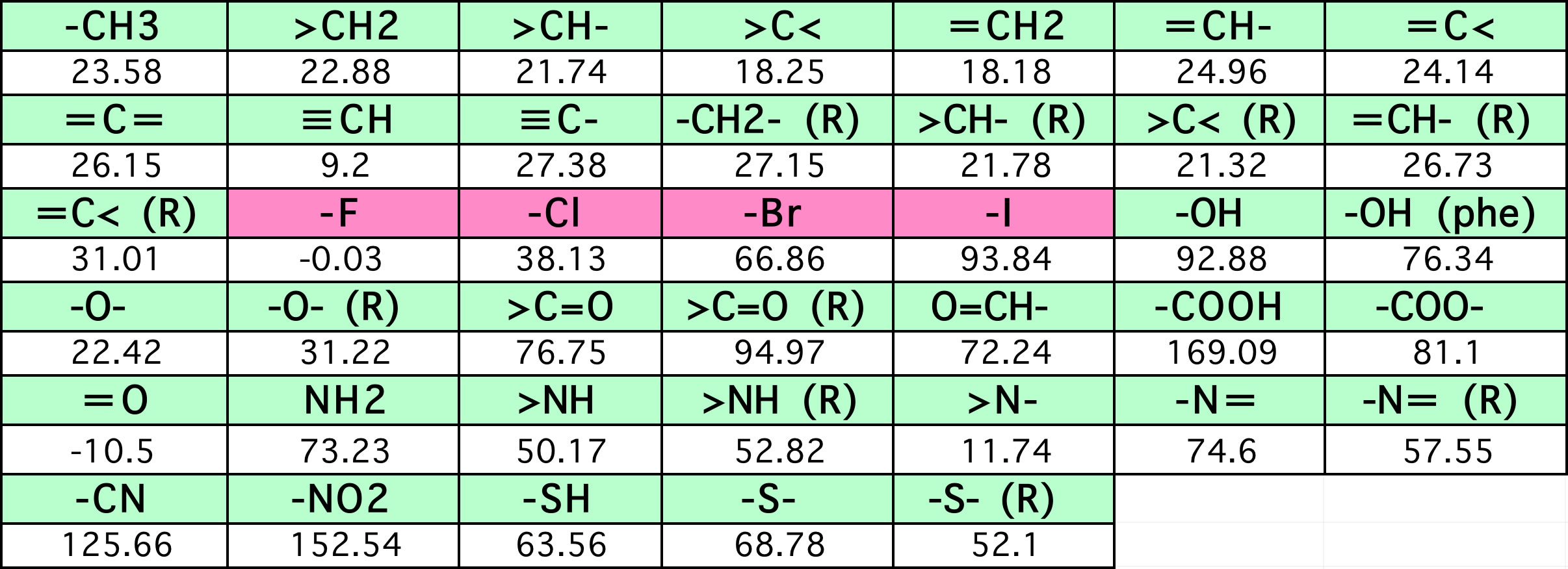
SOLVED: A rudimentary method to calculate the melting temperature of DNA is an equation known as "The Wallace Rule" This equation is: Tm = 2(A+T) + 4(G+C) Where A = # of

Melting Point Examples, Calculation & Range | What is the Melting Point of a Substance? - Video & Lesson Transcript | Study.com

On prediction of melting points without computer simulation: A focus on energetic molecular crystals - ScienceDirect

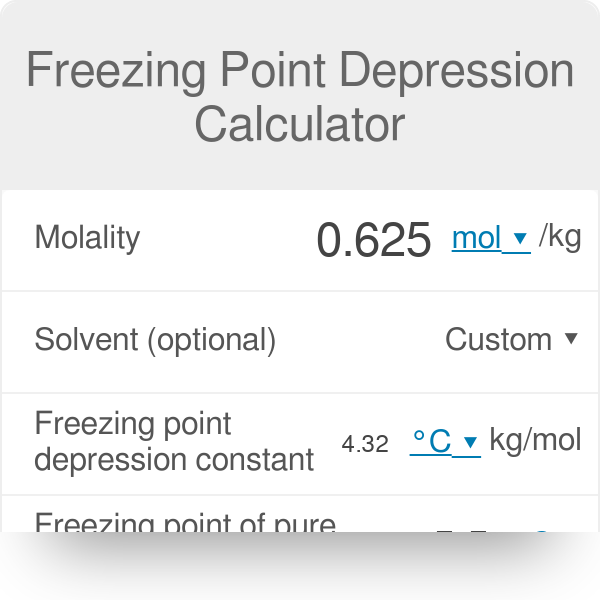
Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube

A fully automated approach to calculate the melting temperature of elemental crystals - ScienceDirect
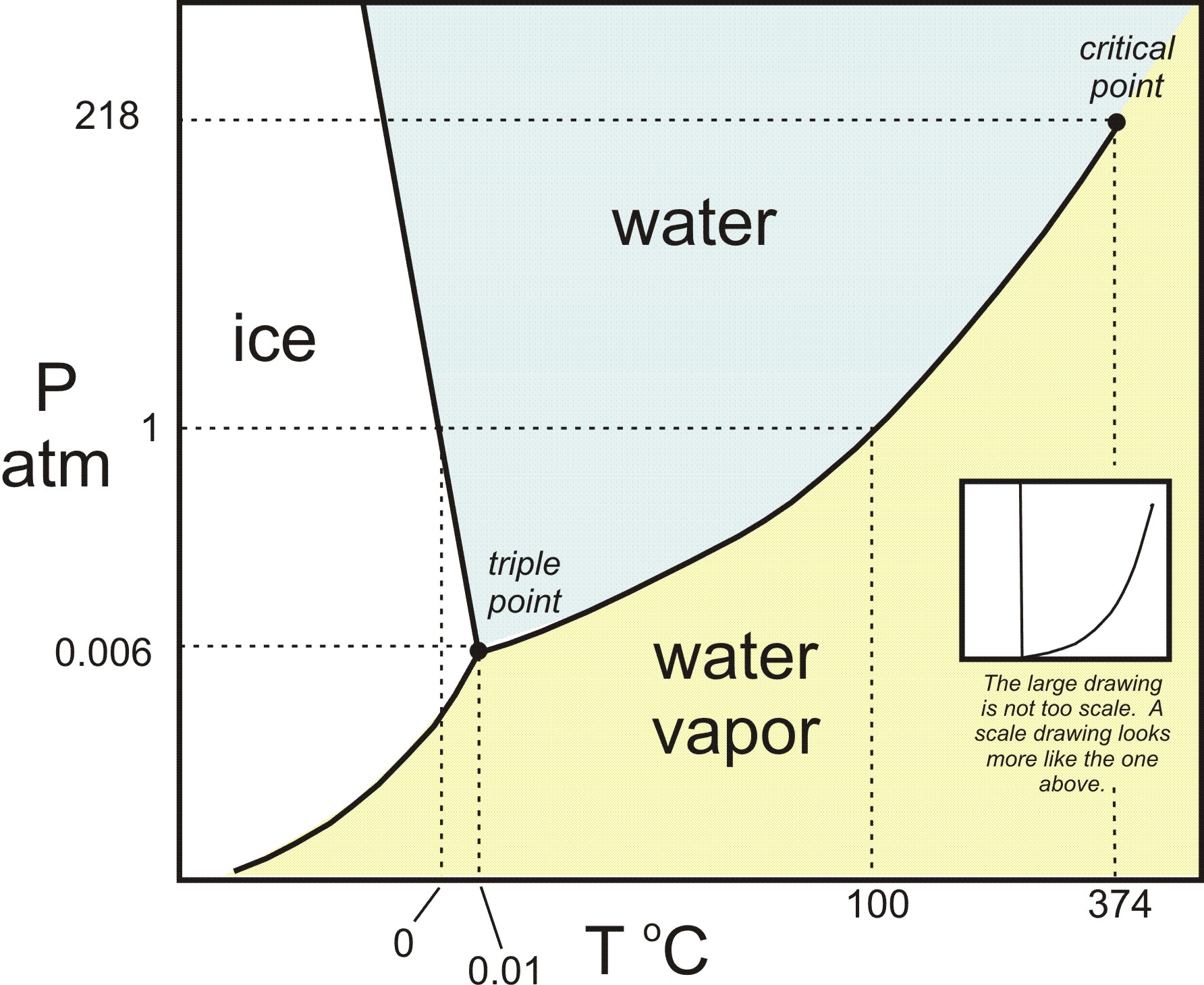
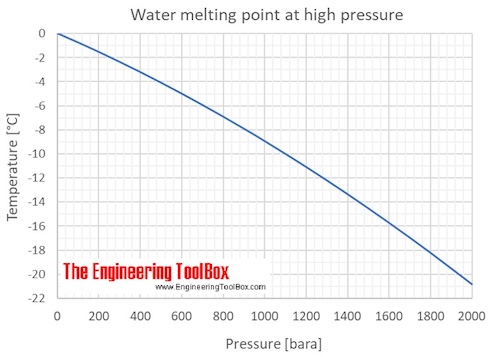
Calculate the change in freezing point for ice if the pressure changes from "1 atm" to "3 atm"? | Socratic